GCSE Science | Electrolysis

How do we separate metals?
Use carbon
If the metal is less reactive than carbon, we can react them with carbon.
Copper oxide + carbon → __________ + __________
Iron(III) oxide + carbon → __________ + __________
Electrolysis
If the metal is more reactive than carbon, we can use electrolysis.
What is electrolysis?
Electrolysis is used to separate a metal from a non-metal.
What is cryolite?
Cryolite is added to lower the melting point of the ionic compound.
This means less energy is needed to melt it.
Electrolysis of molten sodium chloride (NaCl)
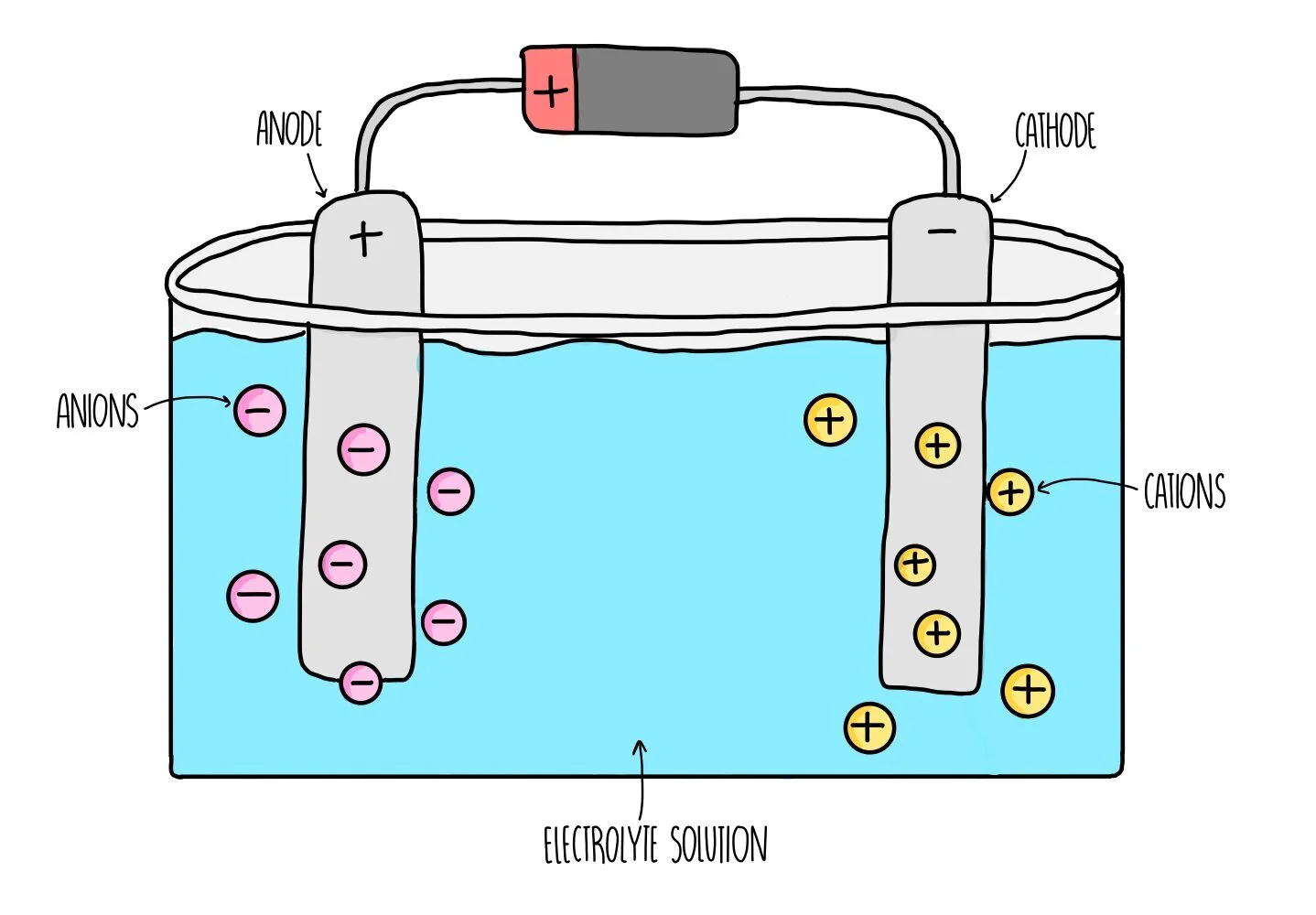
- sodium ions are attracted to the negative electrode
- chlorine ions are attracted to the positive electrode
Half equations

What is an aqeous solution?
An aqeous solution has been dissolved in water.
This means it also contains H+ and OH- ions.
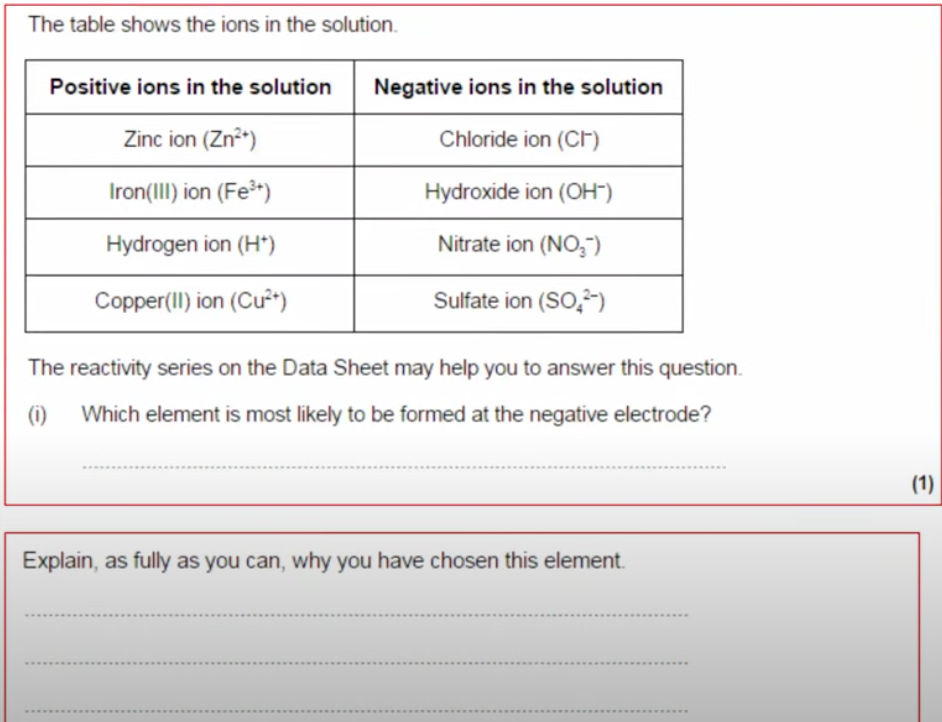
What goes to the positive electrode?
The negative ion goes to the positive electrode.
If there are two negative ions, the halide ion (group 7) is prioritised.
What goes to the negative electrode?
The positive ion goes to the negative electrode.
If there are two positive ions, the one that is less reactive is prioritised.
Practice questions
Question 1
Question 2
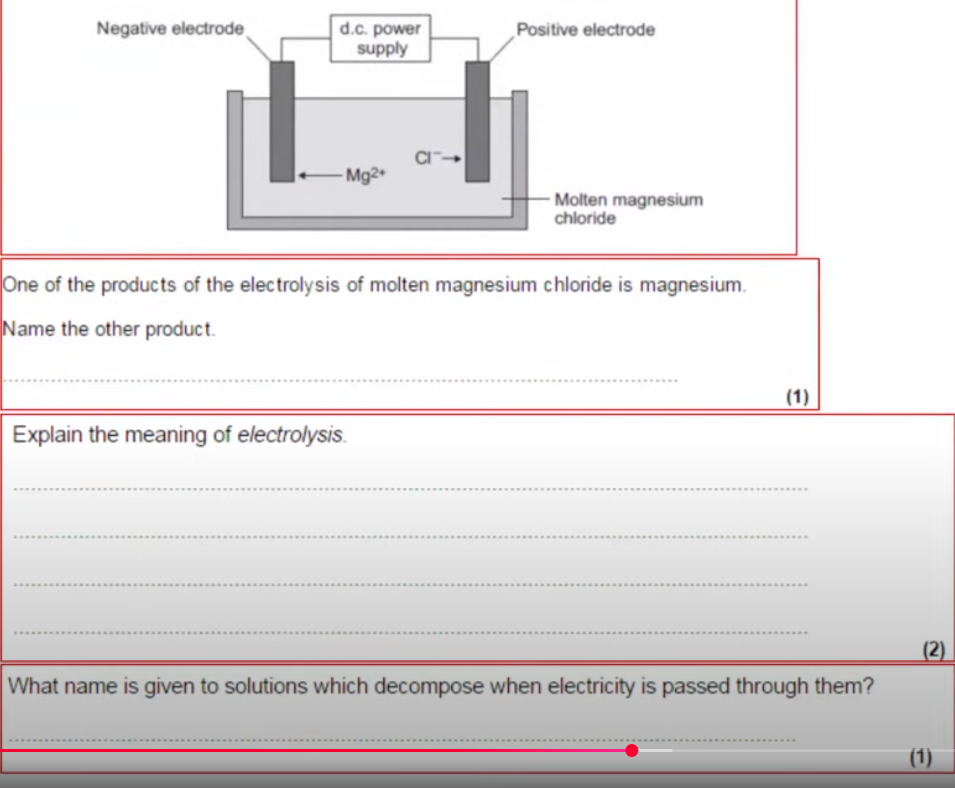
Question 3

Question 4
Question 5
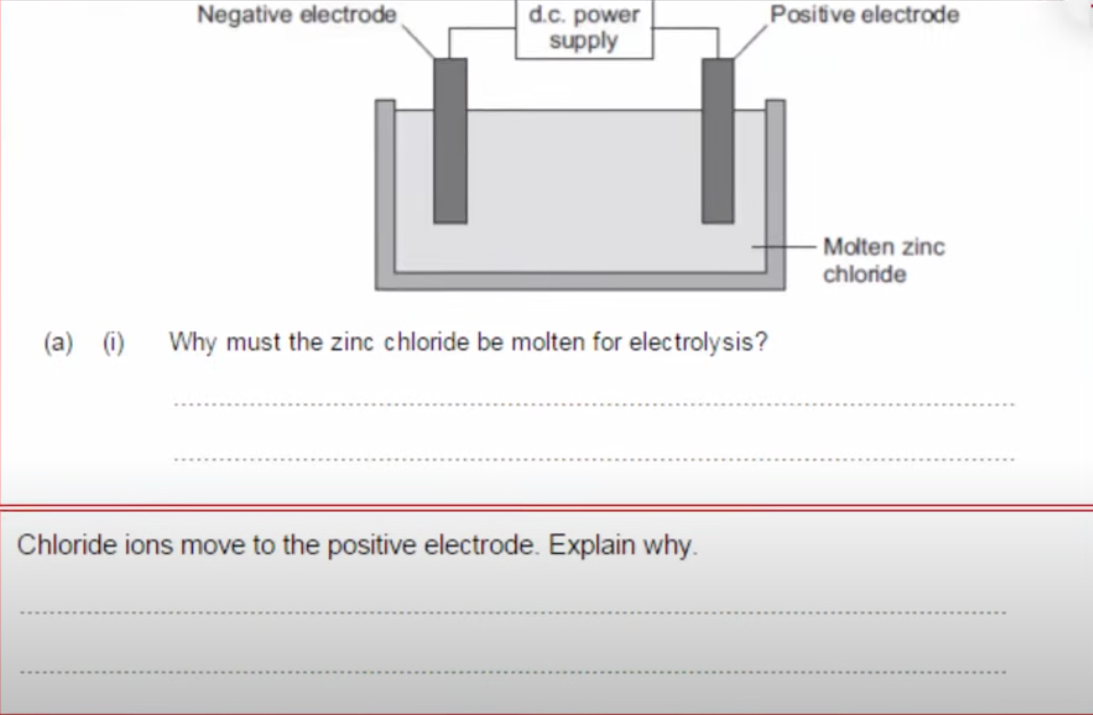
Question 6

Question 7